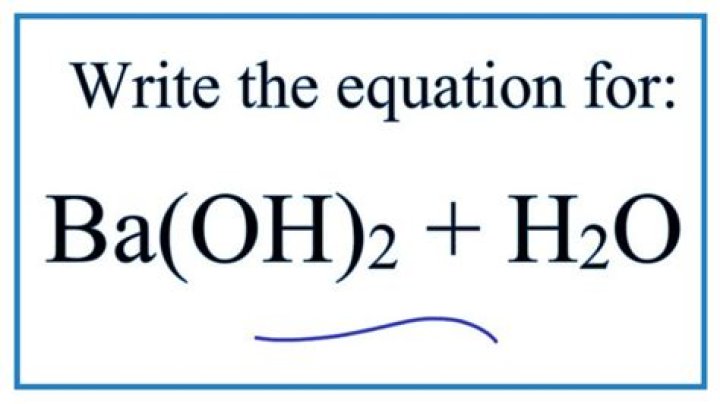
Ba Oh 2 Acid Or Base
 Christopher Ramos
Christopher Ramos BA OH 2 is a strong or weak base?
The solubility of Ba (OH) 2 is much higher than that of Sr (OH) 2. Therefore, Ba (OH) 2 can give more OH to the aqueous solution. Therefore, Ba (OH) 2 is a stronger base than Sr (OH) 2.
Of these, is BA OH 2 a base or an acid?
Answer: Ba (OH) 2 dissociates OH, which can absorb H +. Therefore, Ba (OH) 2 is a base. In an aqueous solution, anything that results in OH is considered a base.
Is FrOH also a solid foundation?
If FrOH could be produced (Fr is radioactive with a short half-life, so there is essentially nothing that exists on earth except as a radioactive degradation product of radium) it would be a strong base.
We also ask ourselves: is BA OH 2 a weak acid?
Common examples of strong Arrhenius bases are alkali metal hydroxides and alkaline earth metals such as NaOH and Ca (OH) 2. Strong bases can deprotonate weak acids, very strong bases can deprotonate very strong, weakly acidic CH groups in absence of water.
Barium hydroxide (Ba (OH) 2)What are the 8 strong foundations?
Here is a list of the most common strong foundations.
- Lithium hydroxide LiOH.
- Sodium hydroxide NaOH.
- Potassium hydroxide KOH.
- Rubidium hydroxide RbOH.
- Cesium hydroxide CsOH.
- * Calcium hydroxide Ca (OH) 2.
- * Sr (OH) 2 strontium hydroxide.
- * Ba (OH) 2 barium hydroxide.
How do you recognize the strong foundations?
A strong base is something like sodium hydroxide or potassium hydroxide, which is completely ionic. You can think of the compound as being 100% divided into metal ions and hydroxide ions in solution. Each mole of sodium hydroxide dissolves to give one mole of hydroxide ions in solution.
What is the strongest foundation?
The hydroxide ion is the strongest possible base in aqueous solutions, but there are bases with much greater powers than those in water. Superbases are valuable in organic synthesis and of fundamental importance in physical organic chemistry. Superbases have been described and used since 1850.
What are the 7 strong acids?
There are 7 strong acids: hydrochloric acid, hydrochloric acid, hydrochloric acid, hydrochloric acid, nitric acid, perchloric acid and sulfuric acid. Inclusion in the list of strong acids does not provide any indication of the dangerousness or harmfulness of an acid.
NaOH is stronger than BA Oh 2?
KOH can give more OH anions than barium hydroxide because the polarization power of the K cation is lower than that of the Ba cation, since the polarization power depends on the charge of the cation and Ba has a +2 charge and K has a +1 charge . Due to its high polarization capacity, the Ba cation therefore attracts more OH anions than the K cation.
Is NaOH basic or KOH?
Is Koh a weak base?
What makes a base weak?
A weak base is a chemical base that does not completely ionize in aqueous solution. Since Brønsted Lowry bases are proton acceptors, a weak base can also be defined as an incomplete protonation chemical base.
Is NaCl an acid or a base?
NaCl is created by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react with each other, salt and water are formed. Therefore, NaCl is a salt.
Is NaCl an electrolyte?
NaCl is an ionic compound. Since it dissolves completely and forms Na + and Cl ions, it is classified as a strong electrolyte. Strong electrolytes in solution are completely dissociated into ions and strongly conduct electricity, i.e. they ionize completely in water and are good conductors of electricity.
What is the strongest electrolyte?
Classification of Electrolytes
Is Sugar an Electrolyte?
Is HCl a strong acid?
A strong acid is one that is fully ionized in an aqueous solution. Hydrochloric acid (HCl) completely ionizes in water to hydrogen ions and chloride ions. Since HCl is a strong acid, its conjugate base (Cl-) is extremely weak.
Is HF stronger than HCl?
HCl is a stronger acid than HF. A molecule that produces several protons in one solution is more acidic. Given the differences in electronegativity, the HF bond should be weaker and therefore easily cleaved to give more H + in a solution, making the HF a stronger acid.
Is water a strong electrolyte?
Is H2O an electrolyte or a non-electrolyte?
Ultrapure water has an ion concentration in the order of magnitude of a very low ion concentration. Since ions conduct electricity, ultrapure water has extremely low electrical conductivity. It is NOT an electrolyte.
Is H3po4 a strong or weak acid?
With the higher electronegativity from N to P, the extra oxygen makes HNO3 a strong acid, while its absence makes H3PO4 weak. After H3PO4 has lost a proton, it forms H2PO4, which has insufficient resonance stabilization. The more resonant structures a molecule has, the more stable it is.
Is Koh stronger than NaOH?
What happens if you mix two acids together?
There is no reaction when two acids are mixed, but acids can react with other acids. Acid-base reactions are possible between a strong acid and a weak acid (which acts as a base), but usually occur with weak acids, which are often not referred to as acids.