Cabr2 Lewis Structure - How To Discuss
 Sophia Dalton
Sophia Dalton Cabr2 Lewis Structure
How to draw the structure of Lewis CaBr2 point? 3
Lanceo de Wolverba (TV), Italy
You ask: How to draw the structure of Lewis CaBr2 point?
Lewis, chemist
Gilbert Ann Lewis was an American chemist born in the late 19th century and lived in Calais until the 1930s.
His contribution to the development of chemistry was immense.
You may remember LEWIS CUC models and DRA UT OCTET ATOMIC VALENCIA.
You may remember your UT chemical link model, such as the chemical link of pure coulomb with atoms, fuel gas chemical elements (excluding gases) and other non-polar molecular compounds such as hydrocarbides.
You need to remember your acid-base theory, which is based on a pair of hydrogen ions alone.
For molecular connections you need to remember your point structure.
Lewis dot diagram
This structure is derived from the atomization of chemical compounds. Therefore, Lewis highlights the solitary pairs of electrons and other external or valence electrons that face the atoms in the structure.
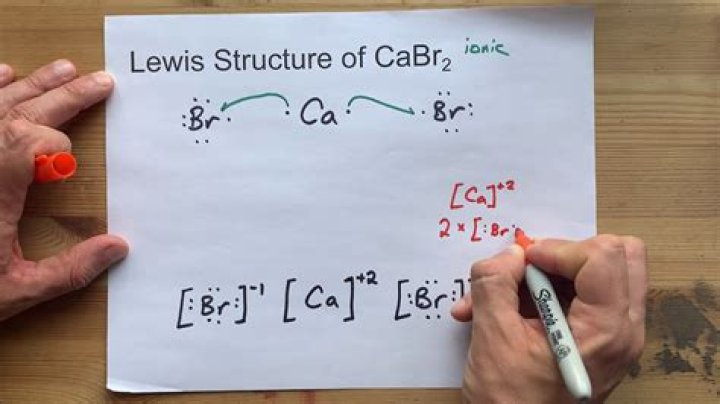
ut CaBr2 (such as calcium bromide)
....
: Br: Ca: Br:
....
You will see that there is no glue for chemical bonds: in fact, the 17 dots around the involved atoms represent the external electrons. Eight electrons around the bromatum.
I can help you.
Cabr2 Lewis Structure
Cabr2 Lewis Structure
Lewis structure of calcium bromide
Ca has 2 electrons, Br has 7. The number of electrons is 16.
Make Ca the central atom. It only makes two connections. Tie Ca to each Br.
BrCaBr
In this process, you used 4 electrons, so you have 12. To use the remaining electrons, place 6 dots around each Br. This is the correct Lewis structure.
Two up, two sides and two down. Your Lewis structure.