Heat and pressure - How To Discuss
 Sophia Dalton
Sophia Dalton Heat and pressure
What is the process of heat and pressure? Heat can result from compression gravity, friction between tectonic plates, or radioactivity. The pressure increases as the rock sinks into the ground and more rocks accumulate in it. There is also direct pressure from plate tectonics. Chemical liquids such as water circulate when rocks are heated and ions are exchanged between the liquid and the rocks.
Does heat and pressure create a metamorphic rock?
Metamorphic rocks form when heat and pressure convert existing rock into new rock. Contact metamorphosis occurs when hot magma transforms the rock it comes in contact with. Regional metamorphoses alter large areas of existing rock under the tremendous heat and pressure created by tectonic forces.
What does heat and pressure change shale to?
Shale exposed to intense heat and pressure can turn into shale. After formation, the shale is usually deposited in lakes and rivers with low water levels. Clay is an important component of slate.
What is specific heat at constant pressure?
Specific heat at constant pressure is the heat applied to the unit mass of the system to raise the temperature by 1 K while keeping the pressure constant.
What is the process of heat and pressure flow
The heat Q Q flows in a monatomic ideal gas and the volume increases while the pressure remains constant. How much of the thermal energy is used for the gas expansion work. The answer from the book is $ \\ frac $ 25.
How is heat transferred from hot to cold fluid?
Devices in which heat is transferred from a hot liquid stream to a cold liquid stream are called heat exchangers. The throttling process occurs when a liquid flowing in a pipe suddenly encounters a restriction in the flow channel. It can be a capillary tube, which is usually in a refrigerator, as shown in figure (c).
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What's the difference between flow, velocity, and pressure?
What's the difference between flow, velocity, and pressure?
Airflow is a measure of airflow in units of volume per unit of time. Common units of measurement are liters per minute, cubic feet per minute (CFM), etc. Velocity is the rate at which air travels a distance per unit of time. Common units are feet per second, meters per second, and so on. Pressure is a measure of the force acting on a surface.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() When is a flow process known as a steady state?
When is a flow process known as a steady state?
When a transmission process meets the following conditions, it is called a constant current. When a transmission process meets the following conditions, it is called a constant current. 1. The mass and energy content of the test volume remains constant over time.
Which is a process modelled on a PV diagram?
All major thermodynamic processes modeled on photoelectric diagrams (isochore, isobaric and isothermal processes) obey the ideal gas law, with the exception of adiabatic processes, which are discussed in detail on the homepage. Below are examples of each process modeled on a photovoltaic diagram.
How is heat added during the expansion process?
Heat must be added during the expansion process. define the enthalpy H of the system by the equation H = U + PV. Enthalpy is a physical property of a system. It only depends on the state of the system (internal energy, pressure, volume) and not on how it got to this state.
How is the internal energy of a gas determined?
Any heat added to the system is used to increase internal energy. The isothermal process takes place at a constant temperature. Since the internal energy of a gas depends only on its temperature, ΔU = applies to the isothermal process. For the isothermal expansion of an ideal gas, W = nRT ln (V 2 / V 1) is used.
What can you do with a pressure volume diagram?
Moreover, these graphs not only simulate the relationship between pressure and volume of an ideal gas, but can also be used to calculate the work done (in a system or system). This is done by calculating the negative area under the curve, which can be done geometrically or by integration.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the process of heat and pressure water
What is the process of heat and pressure water
Warm up water. Water heating is a process of heat transfer that uses an energy source to heat water above its initial temperature. Typical domestic uses for hot water include cooking, cleaning, bathing and space heating. In industry, hot water and steam heated water are used in different ways.
Where does the heat from the water come from?
Thermal energy can come directly from the sun, which heats the water, or from ocean currents, which carry the heated water. Atmospheric pressure is the effect of the pressure of air particles on water. As they descend, a lower pressure pushes the water molecules downward, making it easier for them to rise to the atmosphere.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How is the condensation of water related to temperature?
How is the condensation of water related to temperature?
Condensation is the process by which water vapor becomes liquid again when it cools down. When heat is lost, the water molecules slow down and condense into droplets. This process is mainly influenced by the temperature, but also by the increase in the amount of vapor in the atmosphere.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What happens to water molecules as heat is lost?
What happens to water molecules as heat is lost?
As heat is lost, the water molecules slow down and condense into droplets. This process is mainly influenced by the temperature, but also by the increase in the amount of vapor in the atmosphere. The rising vapor cools and condenses into droplets that adhere to the dust and collect in clouds.
What is the process of heat and pressure in the body
Heat is generated in the body through metabolism, which is a chemical reaction in all cells of the body. Acts that require additional chemical reactions increase metabolism. When metabolism increases, extra heat is released.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How does blood flow to the skin to increase heat loss?
How does blood flow to the skin to increase heat loss?
Peripheral vasodilation increases blood flow to the skin, increasing heat loss. Peripheral vasoconstriction minimizes blood flow to the skin and prevents heat loss. Conduction is the transfer of heat from one object to another through direct contact. Convection is the transfer of heat through the movement of air.
![:brown_circle: :brown_circle:]() How are two specific heats defined in thermodynamics?
How are two specific heats defined in thermodynamics?
Two specific heats are determined for gases: one for constant volume (cv) and one for constant pressure (cp). According to the first law of thermodynamics, the molar specific heat for a constant volume process with a monatomic ideal gas is:
How does the body lose heat through evaporation?
Evaporation is the transfer of thermal energy when a liquid turns into a gas. The body constantly loses heat through evaporation. Every day about 600-900 ml of water evaporates from the skin and lungs, leading to loss of water and heat. By regulating perspiration, the body promotes additional evaporation and heat loss.
What is the process of heat and pressure therapy
Heat and pressure have long been used independently of each other to preserve food. For example, high pressure treatment (HPP) at low or ambient temperatures can prevent the growth of bacterial spores and is an effective means of preserving foods with moderate to high acidity.
What kind of physical therapy does moist heat?
In a moist heat treatment, a warm compress is applied to the tight or painful area and left there for about 10-15 minutes. Other physical therapy treatments include ultrasound, electrical stimulation, and iontophoresis.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How does moist heat help in the healing process?
How does moist heat help in the healing process?
How it speeds up healing. Moist heat also aids the healing process by increasing enzyme activity. This increases metabolism and helps release oxygen from the hemoglobin molecule to tissue, where it aids in recovery.
How does moist heat work for back pain?
Moist heat helps control pain by preventing pain signals from being transmitted. Warming up can also help relieve pain by reducing muscle spasms and promoting healing (see below for more on this). While opinions differ on this, many experts recommend using moist heat only for chronic or sub-acute back or neck pain.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Why are moist heat packs used for burn treatment?
Why are moist heat packs used for burn treatment?
When the moist heat packs are ready for use, they are placed in special towels and/or envelopes. This not only prevents burns, but also slows down the cooling process so you have more time to enjoy the treatment. And of course it is more hygienic.
![:brown_circle: :brown_circle:]() How are heat and work related in thermodynamics?
How are heat and work related in thermodynamics?
A temperature difference is required to transfer heat between the system and the environment. Tare unit in degrees Celsius or Kelvin. The first law of thermodynamics. Work and heat are two ways of transferring energy between the system and the environment, changing the energy of the system.
![:brown_circle: :brown_circle:]() Why does the amount of heat transfer depend on temperature?
Why does the amount of heat transfer depend on temperature?
Since there is no change in net energy, there is no change in temperature. However, at different temperatures, warmer objects emit more energy as radiation than they absorb, while the reverse is true for cooler objects. The amount of energy radiated by an object is highly dependent on its temperature.
What happens when 400 J of heat is added to a gas?
If 10 ■■■■■ of an ideal monatomic gas is slowly heated to 400 J of heat, its temperature will rise. What do you do with the gas? A mole of dilute diatomic gas occupying volume L expands when heated slowly at constant atmospheric pressure.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How is heat transferred from one body to the other?
How is heat transferred from one body to the other?
Two bodies in thermal contact change their temperature until they reach the same temperature. When thermal equilibrium is reached, heat is transferred from one body to another.
What is the process of heat and pressure related
For gases, the relationship between pressure and temperature is described. ■■■-Lussac's law is a gas law that describes the relationship between pressure and temperature. This means that at a constant volume, the pressure of a given amount of a given gas is directly proportional to its Kelvin temperature.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How is heat related to pressure and temperature?
How is heat related to pressure and temperature?
This is the result of a long historical development. Heat is the mixing energy of the elementary particles that make up matter: atomic molecules and electrons. In the case of gases, a simple application of the laws of mechanics makes it possible to establish the ideal gas law in relation to pressure, volume and temperature.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How are pressure, temperature and heat used in everyday life?
How are pressure, temperature and heat used in everyday life?
Pressure, temperature and heat are variable factors in everyday life, especially in meteorology. However, its physical definition is more difficult than it seems. This is the result of a long historical development. Heat is the mixing energy of the elementary particles that make up matter: atomic molecules and electrons.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which is the result of a temperature difference between two bodies?
Which is the result of a temperature difference between two bodies?
The heat flux is the result of the temperature difference between the two bodies and the heat flux is zero when TS = TE. Heat flow drawing. Heat is not the only way to transfer energy between a system and its environment. Energy can also be transferred between the system and the environment through work (W).
What is the equation for heat absorbed under constant pressure?
Under constant pressure conditions, the equation can be written as follows: dU = q P - = (U 2 + PV 2) - (U 1 + PV 1) ……… (2) q P is the heat absorbed at constant pressure. In other words, the heat absorbed at constant pressure corresponds to an increase in enthalpy or heat content if the work is done with volumetric pressure only.
How does heat transfer depend on the process?
Performance and heat transfer depend on the state change process. For a special case of a constant pressure process, the work of the gas is expressed as constant pressure p multiplied by the change in volume V: Substituting this into the first equation, they get:
![:brown_circle: :brown_circle:]() How is the rate of change of the heat equation determined?
How is the rate of change of the heat equation determined?
This feature changes over time as the heat spreads around the room. The heat equation is used to determine the function u over time. The rate of change of u is proportional to the curvature of u. The sharper the angle, the faster it will round.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How are constant pressure processes defined in thermodynamics?
How are constant pressure processes defined in thermodynamics?
The practical thermodynamic function of enthalpy or heat content is used to describe a process at constant pressure. This function is denoted by the letter H and is defined as H = U + PV ……… (1) Like any other thermodynamic state function, it is impossible to determine the absolute value of the enthalpy of the system.
Does heat and pressure inside the Earth form metamorphic rock?
Metamorphic rocks occur when rocks are exposed to high temperatures, high pressures, hot mineral-rich fluids, or more often a combination of these factors. Similar conditions can be found deep underground or where plate tectonics converge.
![:brown_circle: :brown_circle:]() Does pressure affect metamorphic rock?
Does pressure affect metamorphic rock?
To create a metamorphic rock, it is important that the existing rock remains solid and does not melt. Too high a temperature or pressure causes the rock to melt and turn into magma. This results in the formation of volcanic rock rather than metamorphic rock.
![:brown_circle: :brown_circle:]() How can heat change rock to metamorphic rock?
How can heat change rock to metamorphic rock?
Metamorphic rocks are formed from other rocks that are altered by heat or pressure. They are not made of molten rock, which instead melts into volcanic rock. Ground movements can cause rocks to collapse or crush deeply. This heats the rocks and puts them under high pressure.
What are the heat sources for rock metamorphism?
The second heat source comes from intrusive igneous bodies such as batholiths. When these bodies cool, they give off heat to the surrounding rock, leading to metamorphosis. At the end of the metamorphosis, a fusion takes place. The melting point of the rock varies from a minimum of 700 to 800 degrees C to a maximum of 1000 degrees C.
Does heat and pressure create a metamorphic rock structure
Metamorphic rocks form beneath the Earth's surface. They change under extreme heat and pressure. But when lava cools slowly, a lot of texture appears in the rocks, gas bubbles, small holes and cavities. This type of stone consists of sand, shells, pebbles and other materials.
Does heat and pressure create a metamorphic rock cycle
Metamorphic rocks occur when rocks are exposed to high temperatures, high pressures, hot mineral-rich fluids, or more often a combination of these factors. Similar conditions can be found deep underground or where plate tectonics converge.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Does heat and pressure create a metamorphic rock definition
Does heat and pressure create a metamorphic rock definition
Metamorphic rocks are created by transforming existing rocks in a process called metamorphosis, which means change of shape. The original rock (protolith) is exposed to heat (temperatures above 150-200°C) and pressure (100 megapascals (1000 bar) or more), causing profound physical or chemical changes.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Does heat and pressure create a metamorphic rock in order
Does heat and pressure create a metamorphic rock in order
These conditions are usually found deep in the Earth's crust or at the edges of plates where tectonic plates collide. To create a metamorphic rock, it is important that the existing rock remains solid and does not melt. Too high a temperature or pressure causes the rock to melt and turn into magma.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Does heat and pressure create a metamorphic rock made
Does heat and pressure create a metamorphic rock made
When exposed to intense heat and pressure, granite turns into a metamorphic rock called gneiss. Shale is another common metamorphic rock that forms from shale. Limestone, a sedimentary rock, turns into metamorphic stone marble when the necessary conditions are met.
Does heat and pressure create a metamorphic rock type
Metamorphic rocks are formed by heat and pressure and transform the original rock or host rock into a completely new rock. The host rock can be sedimentary, igneous, or even some other metamorphic rock. The word "metamorphic" comes from Greek and means change of form.
![:brown_circle: :brown_circle:]() How does shale rock form in the ocean?
How does shale rock form in the ocean?
How the slate is made. Shale forms as a result of compaction, usually from particles in slow or calm water, such as river deltas, lakes, swamps or the ocean floor. The heavier particles sink and eventually form sandstone and limestone, while clay and fine silt float on the water.
![:brown_circle: :brown_circle:]() What kind of pressure causes foliated rocks to form?
What kind of pressure causes foliated rocks to form?
Plates are the result of strong pressure (and sometimes, to a lesser extent, heat). When exposed to increasing pressure and heating, shale shows varying degrees of metamorphosis: first it becomes shale, then phyllite, then shale, and finally gneiss.
What makes up the composition of oil shale?
Shale is made up of at least 30 percent clay with varying amounts of quartz, feldspar, carbonates, iron oxides and organic matter. Oil shale, or oil shale, also contains kerogen, a mixture of hydrocarbons extracted from ■■■■ plants and animals.
How big are the particles in shale rock?
Occasionally traces of animals, fossils or even traces of raindrops have been preserved in the shale layers. The debris or clay particles in the shale are less than a few millimeters in diameter, so the rock structure only becomes visible when magnified.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is rock cycle processes involves heat and pressure?
What is rock cycle processes involves heat and pressure?
A rock undergoes a metamorphosis when it is exposed to intense heat and pressure on the Earth's crust. During the metamorphosis, the rock does not melt completely. Rock changes with heat and pressure. Metamorphic rock may have a new mineral composition and/or texture. The rock cycle really has no beginning and no end. Just keep going.
How does pressure affect the rock cycle?
When the stones are ■■■■■■ very deeply, they are in a very hot environment with high pressure. The crystals and texture of rocks change when they turn into metamorphic rocks such as marble or shale. When rocks deep underground are subjected to excessive pressure and temperature, they melt and form a molten rock called magma.
![:brown_circle: :brown_circle:]() What does rock change with heat and pressure?
What does rock change with heat and pressure?
When a rock in the earth is subjected to intense heat and pressure, but does not melt, the rock is transformed. Metamorphoses can change the mineral composition and texture of the rock. For this reason, metamorphic rock may have a new mineral composition and/or texture.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What role does heat play in the rock cycle?
What role does heat play in the rock cycle?
Two major energy sources for the rock cycle are also shown: the sun provides energy for surface processes such as weathering, erosion and transport, and the Earth's internal heat provides energy for processes such as subduction, fusion and metamorphosis.
What does heat and pressure change shale to slate
Shale, subjected to the heat and pressure of metamorphism, becomes a hard, fissile metamorphic rock known as shale. With a steady increase in the degree of metamorphosis, the order is phyllite, then schist, and finally gneiss. What happens when the blackboard becomes a blackboard?
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What causes shale to change into slate rock?
What causes shale to change into slate rock?
What makes oil shale oil shale? Heat and pressure. Shale is a sedimentary rock. It is slightly porous and smooth. Slate is a hard, semi-impermeable metamorphic rock. The pressure compresses the slate and hardens the heat.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What do heat and pressure change shale to?
What do heat and pressure change shale to?
Initially it became shale, but as the temperature and pressure increased it eventually became phyllite, then shale and finally gneiss. Q: How do heat and pressure affect oil shale?
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What kind of rock changes under heat and pressure?
What kind of rock changes under heat and pressure?
When exposed to intense heat and pressure, granite turns into a metamorphic rock called gneiss. Shale is another common metamorphic rock that forms from shale. Limestone, a sedimentary rock, turns into metamorphic stone marble when the necessary conditions are met. READ: How tall is Eddie Cochran?
What kind of rock is slate formed from?
When sediment dives deep, it experiences heat and pressure. This causes the rock to deform and turn into a metamorphic rock. What stone is slate made of? Shale is a metamorphic rock that forms when shale and clay are pressurized and heated in the earth over millions of years.
How are heat and pressure used to Change Rocks?
Heat and pressure are forms of energy, and when existing rock is heated, fluids already present in the rock may also be involved in changing the rock. Heat and pressure are the main drivers of change, but only one of them, heat or pressure, can dominate. Some varieties are mainly pressure modified and these rocks are said to be modified.
How is a metamorphic rock changed by heat?
Metamorphosis literally means "change of shape" and metamorphic rocks can be greatly altered by the heat and pressure of the earth. Any existing rock can deform and transform into a new rock, called a metamorphic rock. Metamorphic rocks do not melt when they form (this would be an eruption) and the changes take place in the solid state.
What causes a rock to change its shape?
Another factor that can transform rocks is the pressure created by tons of other rocks leaning against them from above. Heat and pressure generally work together to resist rocks beneath the Earth's surface.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How does the rock cycle affect the Earth's crust?
How does the rock cycle affect the Earth's crust?
The mountain cycle is a series of processes that create and transform types of rocks in the Earth's crust. This happens when water penetrates the Earth's crust, alters the rock, dissolves some of its minerals and transports it to other places. These dissolved minerals precipitate when the water evaporates.
Heat and pressure on rocks
Heat and pressure. Rearrange atoms in rocks to form larger and heavier minerals. The combination of heat and pressure can cause the minerals in the rock to break down into layers. Metamorphic rocks begin to change at temperatures between 100 and 800 degrees Celsius.
How does heat and pressure change rocks?
One of the main ways to heat (or rather, temperature) and change the pressure in rocks is by dehydrating the minerals that make up them.
What rock is created by intense heat and pressure?
The original rock (protolite) is exposed to heat and pressure, causing profound physical and/or chemical changes. The protolith may be sedimentary, igneous, or other older metamorphic rocks.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What types of rocks are formed by heat and pressure?
What types of rocks are formed by heat and pressure?
Rocks and the rock cycle Metamorphic rocks Metamorphic rocks are formed under the influence of high temperatures and pressures. Igneous rocks Igneous rocks are made up of volcanoes. When a volcano erupts, it spews out a molten rock called magma or lava. Sedimentary rocks Sedimentary rocks are made up of years of sediment that is compressed and solidifies.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is rock altered by pressure and heat?
What is rock altered by pressure and heat?
Phyllite is a common metamorphic rock found in many parts of the world. This occurs when sedimentary rock is easily ■■■■■■ and worn away by heat and pressure caused by regional metamorphism. These are almost always the boundary environments of convergent plates that comprise the continental lithosphere.
![:brown_circle: :brown_circle:]() What is heat and pressure rock cycle?
What is heat and pressure rock cycle?
The rock cycle is an endless process in which rocks move and change continuously over millions of years. Heat and pressure deep within the Earth can transform rock into another type called metamorphic rock. In extremely hot conditions, sedimentary and metamorphic rocks can become so hot that they turn into magma.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the main different between weathering and erosion?
What is the main different between weathering and erosion?
- Definition. Weathering is a geological process in which resources such as rocks are destroyed by contact with the atmosphere, water or biological organisms.
- Movement. There is no displacement of resources with change, but displacement with erosion followed by displacement to a new location.
- It took a while.
- Types.
- Conclusion.
![:brown_circle: :brown_circle:]() What are the two types of weathering and erosion?
What are the two types of weathering and erosion?
Physically and chemically, there are two types of weathering. How does deforestation accelerate the erosion process? The land is supported by trees and other plants. Deforestation is the process of removing trees from an area and leaving the land without an anchor. This makes the soil more susceptible to erosion.
![:brown_circle: :brown_circle:]() Is weathering the same as an erosion?
Is weathering the same as an erosion?
People often think that weathering and erosion are the same thing. Well, it isn't. The definitions of weathering and erosion are: anything that breaks a rock into smaller rocks. Things that remove broken stone. Erosion usually occurs after weathering. Weathering destroys the rock and erosion gains.
What is the result of the processes of weathering and erosion?
Weathering and erosion are natural geological processes that affect, move, and reshape materials on the Earth's surface, such as rocks and soil. Weathering is the breaking of rocks into smaller fragments that change the size and structure of the rock.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Does specific heat change with pressure?
Does specific heat change with pressure?
Specific heat at constant pressure is the change in specific enthalpy compared to temperature when the pressure is held constant (isobaric process).
Does specific heat change with volume?
Unlike the total heat capacity, the specific heat does not depend on mass or volume. It describes the amount of heat that must be carried to a unit mass of a substance to raise its temperature by one degree Celsius. The unit of specific heat capacity is J / (kg ° C) or the equivalent of J / (kg K).
What is constant specific heat in thermodynamics?
Therefore, the terms "specific heat at constant volume" and "specific heat at constant pressure" are unfortunately inappropriate and are thermodynamic properties of a substance and by definition depend only on its state. These are extremely important values that have been determined experimentally as a function of the thermodynamic state for a large number of simple compressible materials.
What are specific heat values?
Specific heat is defined as the standard constant pressure (usually atmospheric) for materials and is generally stated as 25°C (K). A standard temperature is used because the specific heat is temperature dependent and can vary with temperature values.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is specific heat at constant pressure formula
What is specific heat at constant pressure formula
(h2 h1) = cp * (T2 T1) The specific heat cp is called the specific heat at constant pressure and refers to the universal gas constant from the equation of state. This last equation is used to determine specific enthalpy values for a particular temperature. Enthalpy is used in the equation for the energy of liquids.
What is high specific heat
The high specific heat of a substance means that a large amount of heat is required to raise the temperature of the substance. A relatively constant temperature with no jumps or dips is important for survival, as most organisms need temperatures within a narrow range to survive. What does the specific heat tell you?
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is a substance that has a high specific heat?
What is a substance that has a high specific heat?
Water has the highest specific heat of all common substances, 1 calorie / g ° C = J / g ° C. This ensures the temperature stability of land masses surrounded by water, the temperature stability of the human body, makes it an effective refrigerant and many others advantages.
What does it mean to have a high specific heat capacity?
Its high specific heat means it can store a large amount of thermal energy with little change in mass or temperature.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the substance with the highest specific heat?
What is the substance with the highest specific heat?
Liquid water has one of the highest specific heat capacities of common substances, about 4182 J/(K kg) at 20 °C, while ice just below °C has only 2093 J/(K kg). The specific heat capacities of iron, granite and hydrogen gas are about 449, 790 and 14300 J/(K kg), respectively.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Does water have one of the highest specific heat capacities?
Does water have one of the highest specific heat capacities?
Water has the highest specific heat capacity. Therefore, heating and cooling takes a long time.
![:brown_circle: :brown_circle:]() What is specific heat at constant pressure of air
What is specific heat at constant pressure of air
Specific heat (C) is the amount of heat required to change the temperature of a unit mass of a substance by one degree. Isobaric heat capacity (Cp) is used for air in a system with constant pressure (ΔP = 0). Isochoric specific heat (Cv) is used for air in a closed system at constant volume (= isovolumic or isometric).
How do you calculate specific heat of air?
Once you are familiar with the terms used to calculate heat capacity, you need to know the equation used to determine the heat capacity of a substance. Formula: Cp = Q/mΔT. You can use this formula if you want to know the change in the amount of heat instead of the specific heat.
What is the ratio of specific heat of air?
For low velocity air, the ratio of the specific heat capacities is equal to a number constant. If the specific heat capacity is constant, they speak of an ideal calorific gas, and if the specific heat changes with temperature, they speak of an imperfect calorific gas.
What is the correct formula for determining specific heat?
Once you are familiar with the terms used to calculate heat capacity, you need to know the equation used to determine the heat capacity of a substance. Formula: Cp = Q/mΔT. You can use this formula if you want to know the change in the amount of heat instead of the specific heat.
What determines specific heat?
Factors that determine the specific heat. Temperature and pressure. Two factors that change the specific heat of a material are pressure and temperature. Specific heat is defined as the standard constant pressure (usually atmospheric) for materials and is generally stated as 25°C (K).
What are some examples of specific heat?
Specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. Example: The specific heat of water is 1 calorie / gram ° C = Joule / gram ° C, which is higher than any other common substance.
![:brown_circle: :brown_circle:]() How is specific heat calculated?
How is specific heat calculated?
The formula for specific heat is as follows: c = Q / (m * ΔT) Q is the amount of heat supplied or removed (in joules), m is the mass of the sample and ΔT is the difference between the initial temperature and the final temperature of the monster.
What is specific heat at constant pressure and volume
Two specific heats are determined for gases: one for constant volume (cv) and one for constant pressure (cp). According to the first law of thermodynamics, the molar specific heat for a constant volume process with a monatomic ideal gas is: Cv = 3 / 2R = J / mol K.
![:brown_circle: :brown_circle:]() Does iron have higher specific heat than water?
Does iron have higher specific heat than water?
It is well known that water has a higher specific heat than iron. Now consider equal masses of water and iron, which are initially in thermal equilibrium. Same heat, 30.
What metal has the highest specific heat?
Specific heat. An example of a problem. Lithium has the highest specific heat of all pure metals. The temperature of a g-lithium sample increases in K when the energy J is added to it.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the highest specific heat capacity?
What is the highest specific heat capacity?
The natural substance with the highest specific heat is liquid ammonia with a specific heat of J/g K. Hydrogen has the highest specific heat of 14630.
![:brown_circle: :brown_circle:]() What are some characteristics of specific heat?
What are some characteristics of specific heat?
Specific heat is a property that measures the amount of energy required to raise the temperature of 1 gram of a material by 1 °C. The higher the c value, the more difficult it is to heat the material. Also, substances with a high specific heat capacity have difficulty dissipating heat and metals are easily cooled.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is specific heat at constant pressure for water
What is specific heat at constant pressure for water
The specific heat of a gas at constant pressure is called cp, and the specific heat at constant volume is called cv. In the case of solids, the specificity does not depend on the type of procedure. The unit of specific heat is the specific heat of water: 1 calorie per gram of degrees C = Joule per gram of degrees C.
How do you calculate specific heat of water?
Calculate the specific heat as c = Q / (m * ΔT). In your example, this is equal to c = 63000 J / (5 kg * 3 K) = 4200 J / (kg * K). This is the typical heat capacity of water. If you have trouble with units, you can use a temperature or weight conversion calculator.
What are the uses of the specific heat of water?
Application of a car cooler with specific heat capacity. Water is pumped through the channels in the engine block to absorb heat. Plates. Cookware is made of metal, which has a low specific heat and therefore needs less heat to raise the temperature. Radiant heating. Radiant heaters are still used in cold countries to heat the house. Sea breeze, land breeze.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the formula to find specific heat of water Q?
What is the formula to find specific heat of water Q?
As discussed earlier, specific heat is the ratio of the temperature change of an object to the water. Also the formula: Thermal energy = (mass of an object or substance) × (specific heat) × (temperature change) Q = m × c ×.
How do you calculate specific heat capacity?
To calculate specific heat, you need experimental data in which heat is exchanged between a metal sample and another object while monitoring its temperature. Once you have the data, the formula. #Q = m * c * DeltaT # uses true.
How do you calculate heating capacity?
To find out the heat capacity of something, all you need to do is divide the amount of heat energy released by the change in temperature to determine how much energy is needed per degree. Every material in the world has a different heat capacity.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is the formula for heat capacity?
What is the formula for heat capacity?
- Q
- M
- ΔT
- J / (kg * K) However, it may not be necessary for typical specific calorific values.
- Ice:
- Aluminum:
- Lead:
- Water:
- Steam:
- Granite:
What are the uses of heat?
Heat is the kinetic energy of the vibrating molecules. In addition to its obvious uses, such as cooking and heating homes, it is also the energy source for most of the electricity produced on Earth. Whether the facility runs on fossil fuels, biomass or nuclear fuel, the goal is to boil water to power a steam turbine.
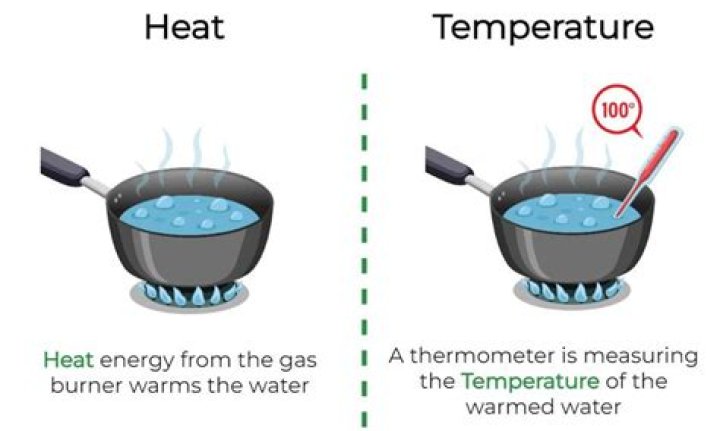
What is heat related to?
Heat is a measure of the number of atoms in a substance multiplied by the amount of energy in each atom. Temperature is related to the speed at which the atoms of a substance move. The temperature of an object is just like the water level, it determines the direction in which the heat flows.
![:brown_circle: :brown_circle:]() What is associated with heat?
What is associated with heat?
Until the mid-19th century, the most commonly used term for heat was a liquid called high-calorie. The eminent chemist Antoine Lavoisier believed that there are two forms of calories: one latent or stored in combustible materials, and the other sensitive and observable when the temperature changes.
What moves from hot to cold?
Hot objects have thermal energy. Thermal energy always changes from hot to colder. Cold energy does not exist, so an object can only cool if thermal energy leaves it. Thermal energy can move in three ways: Conduction. Convection.
Heat and pressure in the rock cycle
Under the influence of heat and pressure, atoms in rocks rearrange to form larger and heavier minerals. The combination of heat and pressure can cause the minerals in the rock to break down into layers. Metamorphic rocks begin to change at temperatures between 100 and 800 degrees Celsius.
Heat and pressure deep beneath earth's surface
Heat and pressure deep in the Earth's surface can transform any rock (igneous, metamorphic, or sedimentary rock) into metamorphic rock. When a rock becomes metamorphic rock, its appearance, texture, crystal structure and mineral composition change.
![:brown_circle: :brown_circle:]() What's the temperature at the bottom of the Earth?
What's the temperature at the bottom of the Earth?
While temperatures three miles deep can be comfortable, at extreme depths you'll encounter infernal temperatures and pressures so intense that ordinary carbon turns into precious diamonds.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What kind of rock can heat and pressure change?
What kind of rock can heat and pressure change?
Heat and pressure deep in the Earth's surface can transform any rock (igneous, metamorphic, or sedimentary rock) into metamorphic rock. When a rock becomes metamorphic rock, its appearance, texture, crystal structure and mineral composition change. B. The bricks are continuously processed in different ways. Right! Thank you.
![:brown_circle: :brown_circle:]() What happens to the earth's temperature when you dig a mile?
What happens to the earth's temperature when you dig a mile?
Geologists have calculated that for every mile dug beneath the Earth's surface, the temperature will rise by 15°F and the pressure will increase at a rate of about 7,300 psi. Violations of the 15 degrees per mile rule are unknown and constitute a notorious no-go zone.
What's the rate at which the earth's pressure increases?
Geologists have calculated that for every mile dug beneath the Earth's surface, the temperature will rise 15°F and the pressure will rise at a rate of about 7,300 psi.
When does heat and pressure change one type of rock?
Aiden 09.09.2020 Metamorphic rocks are formed by heat and pressure and transform the original rock or host rock into a completely new rock. The host rock can be sedimentary, igneous, or even some other metamorphic rock.
Is it true that rocks change over time?
This may be true, but only because their lifespans are so short compared to Earth's history. If you go back and look at geologic time (which focuses on changes over millions of years), you'll see that the rocks actually change! In fact, all breeds change slowly and over and over from one type to another.
![:brown_circle: :brown_circle:]() How does a gneiss form in a rock?
How does a gneiss form in a rock?
Segregation of mineral species often occurs with increasing pressure, and stony gneiss, composed of light and dark mineral bands, can form as the shale changes further as a result of metamorphosis. This can be the result of a gradual change in the shale, but also an increase in the metamorphosis of other rocks.
What kind of rocks form from heat and pressure?
Metamorphic rocks are formed under extreme temperatures or pressure. Unlike other types of rocks, metamorphic rocks start out as rocks, which later turn into different rocks.
![:brown_circle: :brown_circle:]() How do rocks form by heat and pressure?
How do rocks form by heat and pressure?
Rearrange atoms in rocks to form larger and heavier minerals. The combination of heat and pressure can cause the minerals in the rock to break down into layers. Metamorphic rocks begin to change at temperatures between 100 and 800 degrees Celsius. If a rock is compressed and heated over several million years, it can become a new type of rock.