Lewis Structure For Mg - How To Discuss
 William Smith
William Smith Lewis Structure For Mg
What is the Lewis structure of Mg 2 + and S 2? 3
Plot Spread Lewis ...
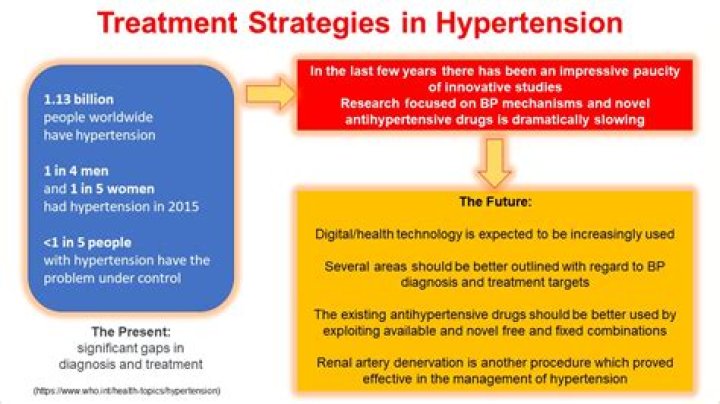
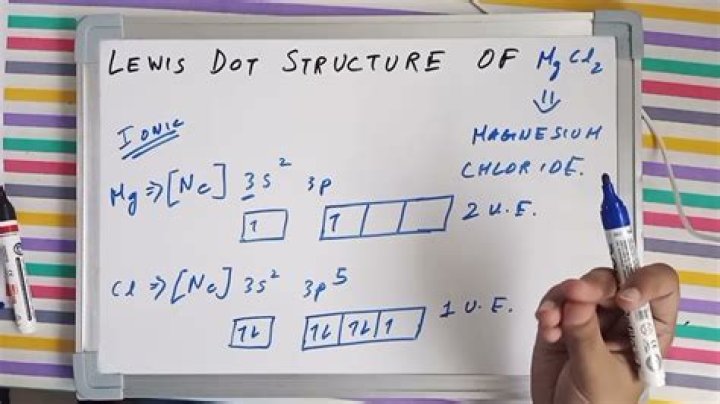
Mg2 + Mg is no more points (because it has lost its two valence electrons)
S2 S plus 8 points (because there are two Mg of six valence electrons accumulated in it)
Lewis point for Mg2 + is meaningless for Mg because there are no electrons in the open s. Basically, Mg2 + is isoelectronic (has the same electronic configuration) as neon. This is a closed s. S2 will be isoelectronic with ■■■■■. You both have closed s, but you still draw it as S which has 8 electrons around it. If you get bad grades, your teacher is wrong. So if you have the MgS compound, all the electrons will be around S for the Lewis dot structure.
This page can help you.
D:
What is the Lewis structure of Mg 2 + and S 2?
Lewis Structure For Mg
Lewis Structure For Mg
Magnesium