Sif5 Lewis Structure - How To Discuss
 Olivia Bennett
Olivia Bennett Sif5 Lewis Structure
Can I get help with some of the Lewis Point structures? 3
I'm having trouble with some of them and I just wanted to review my work. Thank you very much!
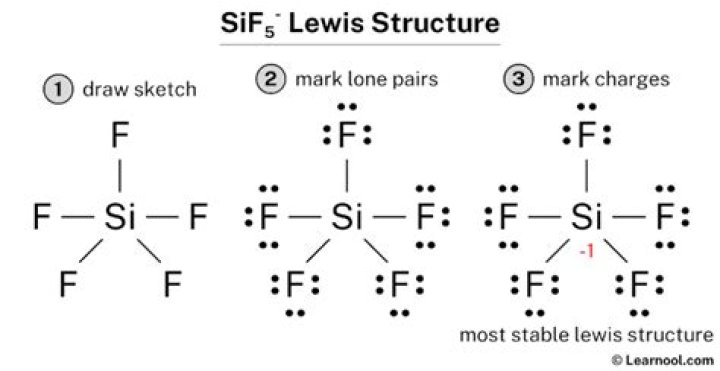
[SiF5] 1
SeO2
updateYes, geometry will help too! Thank you very much!
I think you ...
If as a central atom each F has one bond and each F has 3 pairs of electrons.
It is like a central atom with a pair of electrons, a bond from O (a third pair of this type) and a double bond from the other O (second pair above).
Should geometry also be explained?
Triangular and curved pyramids. . . I'm sorry if it's too late to help.
Lewis Safe5. Structure
This page can help you.
D:
Can I get help with some of the Lewis Point structures?
I'm having trouble with some of them and I just wanted to review my work. Thank you very much!
[SiF5] 1
SeO2
Sif5 Lewis Structure
Sif5 Lewis Structure
Well, fluorine has 7 valence electrons, so any group of 15 elements is fine, but since it cannot be a metal, it must be Nitrogen or Psprus. This is because the charge of group 15 is 3, so the valence electrons are 5 (83 = 5).
I can't help with the first one, but in the second one below:
O = SeO
Well if you move the xs to lol when I see that they all fall to the left. One passes through the first oxygen, the second through Se, and the third through the second oxygen.
The soil is under second oxygen :)
Sif5 Lewis Structure
Sif5 Lewis Structure
Oh, I hate anything with Lewis.com structure and chemistry. I am fine