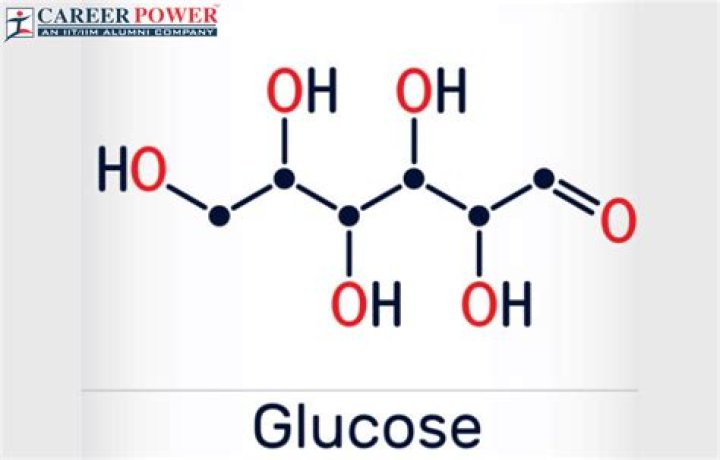
What Mass Of Glucose Should Be Dissolved
 William Smith
William Smith What Mass Of Glucose Should Be Dissolved
How many meters of glucose (C6H12O6) must be dissolved in 10.0 kg of water to get solution with freezing point of 4.2 C? 3
The equation is dT = Kfm.
Kf is the specific constant of water solution Kf is equal to 1.86.
m molarity is m = ■■■■■ / kg solvent of solution
dT, temperature change is basically the freezing point.
By entering a specific quote, you will get:
4.2 = 1.86 meters
m = ....... (value)
From the molarity equation, you know that
m = kg of soluble ■■■■■ / solvent
Water is its solution. You dissolve glucose in it.
Replace the value of m you get in the equation with the kilogram of water you get.
Find the solution (glucose). With the glucose formula, you can calculate the molarity of finding m for glucose.
(0 (4.2)) ° C / (1.86 ° C / m) = 2,258 m
(2.258 mol / kg) x (10.0 kg) x (180.1559 g C6H12O6 / mol) = 4068 g = 4.0 kg C6H12O6